A. Isolation of Casein and Lactose from Milk
Isolation of Casein
Procedure:
Weigh out 5 grams of powdered non-fat dry milk and dissolve it in 20 mL of warm water in a 100 mL beaker. Bring the temperature of the solution to 55oC (do not exceed) on a hot plate, remove the thermometer, and then add dropwise a solution of 10% acetic acid while stirring with a stirring rod. Do not add the acid too rapidly. Continue the acid addition (slightly less than 2 mL will be required), keeping the beaker on the hot plate, until the liquid changes from milky to almost clear and the casein no longer separates. It is important not to add too much acid, because it may hydrolyze some of the lactose in the milk and reduce your yield in Experiment 11B. Stir the precipitated casein until it forms a large amophous mass; then remove it with a stirring rod or tongs and place it in another beaker.
Immediately add 0.75 grams calcium carbonate to the original beaker containing the remaining liquid, stir for a few minutes, and save the resulting mixture for the later separation of lactose below. The separation of lactose should be done as soon as possible during the same laboratory period.
Collect the casein by suction filtration to remove as much water as possible. Press the solid with a spatula. Place the casein in a 100 mL beaker and add 5 mL of a mixture of 1:1 ethyl ether and ethanol (CAUTION: HIGHLY FLAMMABLE - NO FLAMES). Stir the casein in the ether for a few minutes, decant the ether, and repeat the process with a second 5 mL portion of ether. After the second washing with ether, suction filter the product. The ether washings remove any small quantities of fat that may have precipitated with the casein. Place the casein between several layers of paper towels to help dry the product, and let it stand in the air for 10-15 minutes. Divide the wet product in half, and weigh the two portions. Place one portion in a 125 mL Erlenmeyer flask with 35 mL of water and 0.5 mL of 1M NaOH, stopper the mixture, shake it to ensure solution of as much of the casein as possible, and save it for use in the chemical tests below. (You may carry out the chemical tests for the protein during this lab period if you have time, or in your next lab period.) Allow the second portion to dry in your locker over the following two weeks. When dry, weigh this portion and calculate the total yield of casein from the powdered milk. Show your calculations.
Isolation of Lactose
Procedure:
Gently boil the original liquid to which the calcium carbonate was added after isolation of casein. Bumping will not be a problem so long as you stir the solution constantly and vigorously with a glass rod. The solution will foam somewhat as it refluxes. This procedure precipitates the remaining proteins lactalbumin and lactoglobulin. Suction filter the hot mixture to remove the proteins and calcium carbonate, and transfer the hot, slightly yellow filtrate to a 125 mL Erlenmeyer flask. Concentrate the filtrate to a volume of about 5 mL by heating with constant swirling, again being careful to avoid bumping. Foaming can be controlled by heating the liquid less vigorously and gently blowing onto it.
To the hot, concentrated solution, add 25 mL of hot 95% ethanol and 0.2 gram of decolourizing carbon. Put this mixture aside and prepare a slurry of about 1 gram of Celite and 7.5 mL of 95% ethanol. Suction filter the slurry into a Hirsch funnel containing a correct sized filter paper to obtain a filter pad of Celite, and discard the alcohol in the filter flask. [The Celite filter pad helps collect the very fine particles of carbon and prevents the normal filter paper from becoming clogged.]
To the slightly cooled ethanol mixture containing the lactose, add 1 mL water. Suction filter the mixture through the Celite filter pad, making sure the filtrate is clear. If the filtrate is cloudy, heat it up and add another 0.5 mL of water. Transfer the filtrate to a 125 mL Erlenmeyer flask, heat it until it clears, then allow to cool slowly. Stopper the flask and allow it to stand in your locker until your next lab period.
Collect the crystals of lactose by suction filtration, and wash the product with a small amount of cold 95% ethanol. Thoroughly dry the lactose and determine its weight and melting point. Determine the percentage yield of lactose from the powdered milk, and show your calculations.
B. Chemical Tests for Proteins and Carbohydrates
Chemical Tests for Proteins and Amino Acids
In this experiment, you will perform chemical tests on the sample of casein which you isolated from milk, in order to determine the presence of specific amino acids in this type of protein. The tests will also be carried out on the amino acids, to help you identify a positive test with your sample, and on egg albumin, which is the main protein present in egg whites and is similar to the lactalbumin found in milk. While there are literally dozens of tests that are characteristic for only certain amino acids, we will carry out only three.
You will use the aqueous solution of casein which you prepared above (suction filter it if it is cloudy), along with stock solutions of egg albumin, tyrosine, glycine, and cysteine which have been prepared for you. Since some of the reagents used in these tests are toxic and/or corrosive, wear gloves, carry out the FIRST TWO tests in the fume hoods, and dispose of your waste in the proper labelled containers. Since we have more students than fume hoods, you will have to apportion your time carefully and stagger the amino acid tests with the carbohydrate tests, which you can carry out at the bench.
Procedures
1. Millon's Test
Millon's test is given by any compound containing a phenolic hydroxy group. Consequently, any protein containing tyrosine will give a positive test of a pink to dark-red colour. The Millon reagent is a solution of mercuric and mercurous ions in nitric and nitrous acids (CAUTION: MILLON'S REAGENT IS HIGHLY TOXIC AND HIGHLY CORROSIVE). The red colour is probably due to a mercury salt of nitrated tyrosine.
Procedure: Place 1 mL of casein, 2% egg albumin, and 0.1 M tyrosine into separate, labelled, 12 x 75 mm test tubes. Add 3 drops of Millon's reagent and immerse the tubes in a boiling water bath for 5 minutes. Cool the tubes and record the colours formed.
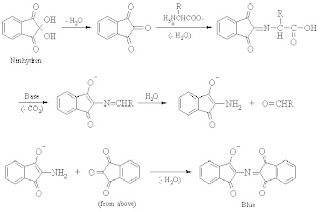
2. Ninhydrin Test
The ninhydrin reaction is used to detect the presence of -amino acids and proteins containing free amino groups. When heated with ninhydrin, these molecules give characteristic deep blue colours (or occasionally pale yellow). The reactions involved in this test are shown below
Procedure:
Place 1 mL of of casein, 2% egg albumin, and 0.1 M glycine into separate, labelled, 12 x 75 mm test tubes. Add 4 drops of 0.1% ninhydrin solution. (CAUTION: NINHYDRIN IS A CARCINOGEN - AVOID DIRECT CONTACT) Add a boiling chip to each test tube and heat to boiling in a hot-water bath. Record the results.
3. Sulfur Test
The presence of sulfur-containing amino acids such as cysteine can be determined by converting the sulfur to an inorganic sulfide through cleavage by base. When the resulting solution is combined with lead acetate, a black precipitate of lead sulfide results.
Sulfur-containing protein ----NaOH----> S2- ----Pb2+----> PbS
Procedure: Place 1 mL of casein, 2% egg albumin, and 0.1 M cysteine into separate, labelled 16 x 150 mm test tubes. Add 2 mL of 10% aqueous sodium hydroxide. Add 5 drops of 10% lead acetate solution. Stopper the tubes and shake them, then remove the stoppers and heat in a boiling water bath for 5 minutes. Cool and record the results.
Chemical Tests for Carbohydrates
In this experiment, you will perform tests and reactions on the sample of lactose which you isolated from milk and on samples of selected other mono- and disaccharides.
1. Benedict's Test
Benedict's test determines whether a monosaccharide or disaccharide is a reducing sugar, and is hence similar in purpose to the Tollens test. To give a positive test, the carbohydrate must contain a hemiacetal which will hydrolyse in aqueous solution to the aldehyde form. Benedict's reagent is an alkaline solution containing cupric ions, which oxidize the aldehyde to a carboxylic acid. In turn, the cupric ions are reduced to cuprous oxide, which forms a red precipitate.
RCHO + 2Cu2+ + 4OH- -----> RCOOH + Cu2O + 2H2O
Procedure Place 15 drops of the following 1% carbohydrate solutions in separate, labelled 12X75 mL test tubes: glucose, fructose, sucrose, lactose, and maltose. Also place 1 mL of distilled water in another tube to serve as a control. To each tube, add 1 mL of Benedict's reagent and heat the tubes in a boiling water bath for 5 minutes. Remove the tubes and note and record the results.
2. Barfoed's Test
Barfoed's test is similar to Benedict's test, but determines if a carbohydrate is a monosaccharide or a disaccharide. Barfoed's reagent reacts with monosaccharides to produce cuprous oxide at a faster rate than disaccharides do:
RCHO + 2Cu2+ + 2H2O -----> RCOOH + Cu2O + 4H+
Procedure: Place 15 drops of the following 1% carbohydrate solutions in separate, labelled 12X75 mL test tubes: glucose, fructose, sucrose, lactose, and maltose. To each tube, add 1 mL of Barfoed's reagent and heat the tubes in a boiling water bath for 10 minutes. Remove the tubes and note and record the results.
3. Hydrolysis Test for Glucose
Disaccharides and polysaccharides can be hydrolyzed in acidic solution into their component monosaccharides, and then submitted to chemical tests like Benedict's test. In this experiment, several disaccharides and a sample of starch will be hydrolyzed, and tested for the presence of glucose. The glucose test will be carried out using a commercially available product called Tes-Tape. Available at most drug stores, the tape contains the enzymes glucose oxidase and peroxidase, as well as ortho-toluidine. The glucose oxidase oxidizes glucose to gluconic acid and hydrogen peroxide. Once formed, the hydrogen peroxide reacts with peroxidase to produce oxygen, which oxidizes the ortho-toluidine to give green-coloured products.
Procedure:
Place 5 mL of the following 1% carbohydrate solutions in separate, labelled 16 x 150mm test tubes: sucrose, lactose, maltose, and starch. Add 3 drops of concentrated hydrochloric acid to each of the tubes, and heat them in a boiling water bath (400 mL beaker) for 10 minutes. Cool the tubes in an ice bath. Carefully neutralize each of the four solutions with 10% sodium hydroxide, using litmus or pH paper. The pH MUST be neutral or very slightly alkaline in order for the Tes-Tape to work. If necessary, make final pH adjustments with 0.1M HCl and/or 0.1M NaOH solutions. Test each solution with Tes-Tape (by placing a drop on the tape and recording the colour change - use plain distilled water as a control) and if time permits, with Benedict's reagent. Record the results and compare them with those obtained earlier with the Benedict's tests on the unhydrolysed carbohydrates.